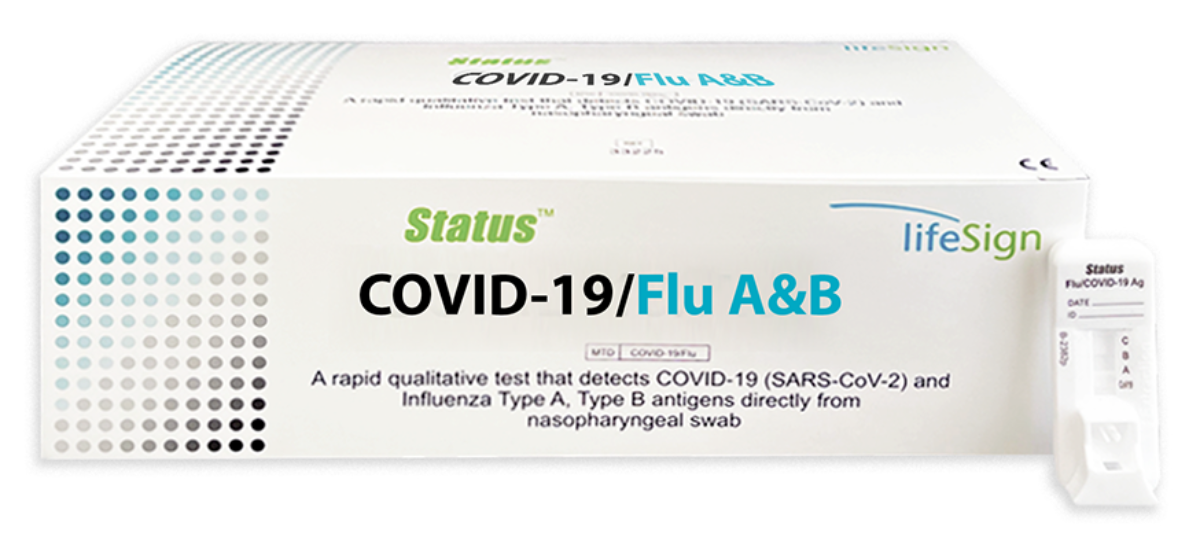
Status- COVID-19/Flu A&B POC Antigen RAPID TEST Save On LLC
$ 500,00 Original price was: $ 500,00.$ 150,00Current price is: $ 150,00.
Description
A Rapid Immunoassay for the Simultaneous Direct Detection and Differential Diagnosis of SARS-CoV-2, Influenza Type A and Type B Antigen from anterior nasal and nasopharyngeal swab specimens
- COVID-19
– Anterior nasal swab specimen − Sensitivity 93.8 %, Specificity 100%
– Nasopharyngeal − Sensitivity 93.1 %, Specificity 100%
- Flu A – Sensitivity 91.4%, Specificity 95.7%
- Flu B – Sensitivity 87.6%, Specificity 95.9%
- FDA Emergency Use Authorization (EUA)
- Visually read in 15 minutes
- Flocked nasopharyngeal swab for superior specimen collection and patient comfort
Features :
- Hygienic Product : Not Specified
- Allergy : Not Specified
Specifications
- UPC:Not Specified
- GTIN/ITF Barcode : Not Specified
- Units of Measure : Not Specified
- Overall Product Length : Not Specified in
- Overall Product Width : Not Specified in
- Overall Product Height : Not Specified in
- Weight Capacity : Not Specified lbs
- Actual Product Weight : 0.80
- Product Assembly : Not Specified
- Product Installation : Not Specified
- Country of Origin : Not Specified
- Case QTY : Not Specified/case
- Case Weight : Not Specified
- Ships Via Freight : No
- Proposition 65 Warning : Not Specified
Quick Shipping and Professional Packaging
We offer a wide range of shipping options due to our long-standing relationships with UPS, FedEx and DHL. Our warehouse staff is highly skilled and will pack your items according to our exact and precise specifications. Your items will undergo an extensive inspection and will be securely secured prior to being delivered. We ship to thousands of customers every day from all over the world. This shows our commitment to being the largest retailer online in the world. We have distribution centers and warehouses in Europe as well as the USA.
Note: Orders containing multiple items will have a separate processing period for each item.
Before shipping, we will examine the items ordered thoroughly. The majority of orders are shipped within 48 hours. Delivery is expected to take between 3 and seven days.
Returns
Stock is dynamic. It is not managed entirely by us, as we have multiple entities, including the factory and the storage. Stocks are subject to change at any time. Be aware that your order is out of stock once your order has been made.
Our policy lasts for 30 days. If you don't receive the product within 30 days, we are not able to provide the option of a refund or exchange.
To be eligible for a return, your item must be in good condition and in the same condition as you received it in. It must also be in the original packaging.
Related products
Save On LLC
Save On LLC
Siemens CLINITEST® Rapid COVID-19 Antigen Self-Test (Pallet of 10,000 tests) Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC
Save On LLC